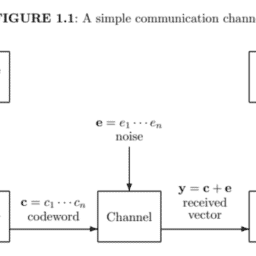
如果你也在 怎样代写信息论information theory 这个学科遇到相关的难题,请随时右上角联系我们的24/7代写客服。信息论information theory回答了通信理论中的两个基本问题:什么是最终的数据压缩(答案:熵$H$),什么是通信的最终传输速率(答案:信道容量$C$)。由于这个原因,一些人认为信息论是通信理论的一个子集。我们认为它远不止于此。
信息论information theory在统计物理学(热力学)、计算机科学(柯尔莫哥洛夫复杂性或算法复杂性)、统计推断(奥卡姆剃刀:“最简单的解释是最好的”)以及概率和统计学(最优假设检验和估计的误差指数)方面都做出了根本性的贡献。
my-assignmentexpert™信息论information theory代写,免费提交作业要求, 满意后付款,成绩80\%以下全额退款,安全省心无顾虑。专业硕 博写手团队,所有订单可靠准时,保证 100% 原创。my-assignmentexpert, 最高质量的信息论information theory作业代写,服务覆盖北美、欧洲、澳洲等 国家。 在代写价格方面,考虑到同学们的经济条件,在保障代写质量的前提下,我们为客户提供最合理的价格。 由于统计Statistics作业种类很多,同时其中的大部分作业在字数上都没有具体要求,因此信息论information theory作业代写的价格不固定。通常在经济学专家查看完作业要求之后会给出报价。作业难度和截止日期对价格也有很大的影响。
想知道您作业确定的价格吗? 免费下单以相关学科的专家能了解具体的要求之后在1-3个小时就提出价格。专家的 报价比上列的价格能便宜好几倍。
my-assignmentexpert™ 为您的留学生涯保驾护航 在澳洲代写方面已经树立了自己的口碑, 保证靠谱, 高质且原创的澳洲代写服务。我们的专家在信息论information theory代写方面经验极为丰富,各种信息论information theory相关的作业也就用不着 说。

数学代写|信息论代写Information Theory代考|The General Behavior of the Probability Density P r(r)
The distribution of momenta determines the contribution of the first two terms on the right hand side of Eq. (2.73) to the entropy of the system. The second distribution $\operatorname{Pr}(r)$ determines the entropy associated with the locations and the interactions among the particles. In this section we explore the dependence of the probability density $\operatorname{Pr}(r)$ on the various parameters of the system.
For ideal gas, $U(r)=0$, and the distribution density reduces to:
$$
\operatorname{Pr}(r)=\frac{\exp [-\beta p r]}{\int_0^{\infty} \exp [-\beta p r] d r}=\frac{\exp [-\beta p r]}{(\beta p)^{-1}}
$$
Since for an ideal gas the equation of state is:
$$
\beta p=\rho=\frac{N}{V}
$$
We can rewrite the probability density as:
$$
\operatorname{Pr}(r)=\rho \exp [-\rho r]
$$
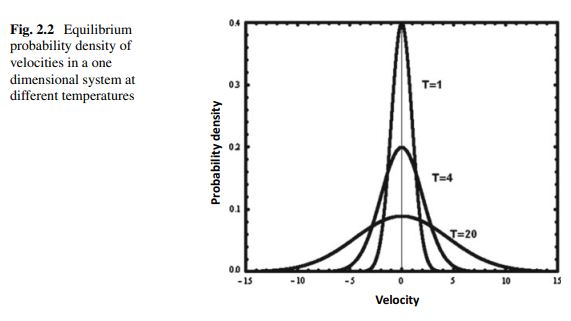
Figure 2.12 shows the probability density $\mathrm{Pr}$ for ideal gas at different densities and at different temperatures $T$, and constant pressure, $P$. In all cases we have an exponential distribution. As expected the distribution becomes sharper as the density increases or as the temperature decreases, at constant pressure. Note that sharper distribution means smaller SMI.
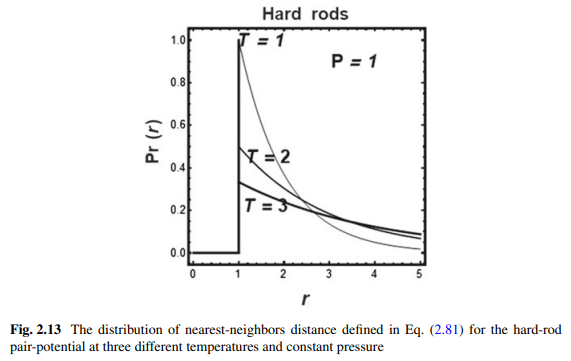
In Fig. 2.13 we show the distribution density for hard rods with diameter $\sigma=1$ and pair potential:
$$
U_{H S}(r)=\left{\begin{array}{l}
\infty, \text { for } r<\sigma \
0, \text { for } r \geq \sigma
\end{array}\right.
$$
The general form of the curves is similar to that of ideal gas. In both cases we note that the distribution becomes more uniform as the temperature increases, or the pressure decreases (or equivalently when the density increases). We will see in the next section that this trend makes the entropy, or the SMI of the system, larger as the density becomes more uniform.
数学代写|信息论代写Information Theory代考|The Entropy-Change Due to Turning-On the Interaction Energy
In this section we study a few examples of the effect of “turning-on” the interaction energy on the entropy of the system. As we already know from the previous sections turning on the interaction energy, at constant $T, P$ always reduces the entropy of the system, or equivalently, increases in the SMI. Before we show a few examples it is important to note that one should be careful in choosing the parameters $p, T$, and $\varepsilon$ of the system (here, $\sigma=\delta=1$ in all of the examples). When the pressure is too high or the temperature too low, the system might reach a close-packed density, Fig. 2.16, beyond which we might get unrealistic values for the entropy-changes, as well as for other thermodynamic quantities. For instance, for hard-rod particles the exact equation of state is:
$$
\beta p=\frac{\rho}{1-\rho \sigma}
$$
Thus, the pressure diverges when $\rho \sigma=1$, for $\rho \sigma>1$ we obtain a negative pressure, which is physically meaningless.
We start with the a system of hard rod particles with interaction energy potential as shown in Fig. 2.7a. For this case we have an exact expression for the chemical potential:
$$
\mu=T \ln (\Lambda p / T)+\sigma p
$$
The change in the chemical potential is simply:
$$
\Delta \mu=\mu-\mu^{i g}=\sigma p
$$
Thus, for the HR of diameter $\sigma=1, \Delta \mu=p$, the corresponding entropy-change is:
$$
\Delta S=S-S^{i g}=0
$$
This result is valid for the process of “turning on” the interaction at $p$ and $T$ constants. However, when the “turning on” of the interactions are carried out at constant volume, or constant density, the change in the entropy is always negative. The exact expression for the change in Gibbs energy per mole of particles at constant $T$ and $\rho$ is:
$$
\Delta \mu=-T \ln (1-\rho \sigma)+\sigma T \frac{\rho}{1-\rho \sigma}
$$
信息论代写
数学代写|信息论代写Information Theory代考|The General Behavior of the Probability Density P r(r)
动量的分布决定了方程(2.73)右侧前两项对系统熵的贡献。第二个分布$\operatorname{Pr}(r)$决定了与位置和粒子间相互作用相关的熵。在本节中,我们将探讨概率密度$\operatorname{Pr}(r)$对系统各种参数的依赖关系。
对于理想气体,$U(r)=0$,分布密度减小为:
$$
\operatorname{Pr}(r)=\frac{\exp [-\beta p r]}{\int_0^{\infty} \exp [-\beta p r] d r}=\frac{\exp [-\beta p r]}{(\beta p)^{-1}}
$$
因为对于理想气体,状态方程为:
$$
\beta p=\rho=\frac{N}{V}
$$
我们可以将概率密度改写为:
$$
\operatorname{Pr}(r)=\rho \exp [-\rho r]
$$
图2.12给出了理想气体在不同密度、不同温度$T$和定压$P$下的概率密度$\mathrm{Pr}$。所有情况下都是指数分布。正如预期的那样,在恒压下,随着密度的增加或温度的降低,分布变得更加明显。请注意,更明显的分布意味着更小的SMI。
如图2.13所示为直径$\sigma=1$、偶电位的硬棒的分布密度:
$$
U_{H S}(r)=\left{\begin{array}{l}
\infty, \text { for } r<\sigma \
0, \text { for } r \geq \sigma
\end{array}\right.
$$
曲线的一般形式与理想气体的曲线相似。在这两种情况下,我们注意到,随着温度的升高或压力的降低(或密度的增加),分布变得更加均匀。我们将在下一节中看到,随着密度变得更加均匀,这种趋势使熵或系统的SMI变得更大。
数学代写|信息论代写Information Theory代考|The Entropy-Change Due to Turning-On the Interaction Energy
在本节中,我们将研究“开启”相互作用能对系统熵的影响的几个例子。正如我们从前面的章节中已经知道的那样,打开相互作用能,在恒定$T, P$下总是减少系统的熵,或者等价地,增加SMI。在我们展示一些示例之前,重要的是要注意,在选择系统的参数$p, T$和$\varepsilon$时应该小心(这里,在所有示例中都是$\sigma=\delta=1$)。当压力过高或温度过低时,系统可能会达到密实密度,如图2.16所示,超过这个密度,我们可能会得到不现实的熵变值,以及其他热力学量。例如,对于硬棒颗粒,精确的状态方程为:
$$
\beta p=\frac{\rho}{1-\rho \sigma}
$$
因此,当$\rho \sigma=1$时压力发散,对于$\rho \sigma>1$,我们得到一个负压,这在物理上是没有意义的。
我们从具有相互作用能势的硬棒粒子系统开始,如图2.7a所示。在这种情况下,我们有化学势的精确表达式:
$$
\mu=T \ln (\Lambda p / T)+\sigma p
$$
化学势的变化很简单:
$$
\Delta \mu=\mu-\mu^{i g}=\sigma p
$$
因此,对于直径$\sigma=1, \Delta \mu=p$的HR,对应的熵变为:
$$
\Delta S=S-S^{i g}=0
$$
该结果适用于在$p$和$T$常数处“打开”相互作用的过程。然而,当相互作用的“开启”在恒定的体积或恒定的密度下进行时,熵的变化总是负的。在$T$和$\rho$恒定时,每摩尔粒子吉布斯能变化的精确表达式为:
$$
\Delta \mu=-T \ln (1-\rho \sigma)+\sigma T \frac{\rho}{1-\rho \sigma}
$$

数学代写|信息论代写Information Theory代考 请认准UprivateTA™. UprivateTA™为您的留学生涯保驾护航。
微观经济学代写
微观经济学是主流经济学的一个分支,研究个人和企业在做出有关稀缺资源分配的决策时的行为以及这些个人和企业之间的相互作用。my-assignmentexpert™ 为您的留学生涯保驾护航 在数学Mathematics作业代写方面已经树立了自己的口碑, 保证靠谱, 高质且原创的数学Mathematics代写服务。我们的专家在图论代写Graph Theory代写方面经验极为丰富,各种图论代写Graph Theory相关的作业也就用不着 说。
线性代数代写
线性代数是数学的一个分支,涉及线性方程,如:线性图,如:以及它们在向量空间和通过矩阵的表示。线性代数是几乎所有数学领域的核心。
博弈论代写
现代博弈论始于约翰-冯-诺伊曼(John von Neumann)提出的两人零和博弈中的混合策略均衡的观点及其证明。冯-诺依曼的原始证明使用了关于连续映射到紧凑凸集的布劳威尔定点定理,这成为博弈论和数学经济学的标准方法。在他的论文之后,1944年,他与奥斯卡-莫根斯特恩(Oskar Morgenstern)共同撰写了《游戏和经济行为理论》一书,该书考虑了几个参与者的合作游戏。这本书的第二版提供了预期效用的公理理论,使数理统计学家和经济学家能够处理不确定性下的决策。
微积分代写
微积分,最初被称为无穷小微积分或 “无穷小的微积分”,是对连续变化的数学研究,就像几何学是对形状的研究,而代数是对算术运算的概括研究一样。
它有两个主要分支,微分和积分;微分涉及瞬时变化率和曲线的斜率,而积分涉及数量的累积,以及曲线下或曲线之间的面积。这两个分支通过微积分的基本定理相互联系,它们利用了无限序列和无限级数收敛到一个明确定义的极限的基本概念 。
计量经济学代写
什么是计量经济学?
计量经济学是统计学和数学模型的定量应用,使用数据来发展理论或测试经济学中的现有假设,并根据历史数据预测未来趋势。它对现实世界的数据进行统计试验,然后将结果与被测试的理论进行比较和对比。
根据你是对测试现有理论感兴趣,还是对利用现有数据在这些观察的基础上提出新的假设感兴趣,计量经济学可以细分为两大类:理论和应用。那些经常从事这种实践的人通常被称为计量经济学家。
Matlab代写
MATLAB 是一种用于技术计算的高性能语言。它将计算、可视化和编程集成在一个易于使用的环境中,其中问题和解决方案以熟悉的数学符号表示。典型用途包括:数学和计算算法开发建模、仿真和原型制作数据分析、探索和可视化科学和工程图形应用程序开发,包括图形用户界面构建MATLAB 是一个交互式系统,其基本数据元素是一个不需要维度的数组。这使您可以解决许多技术计算问题,尤其是那些具有矩阵和向量公式的问题,而只需用 C 或 Fortran 等标量非交互式语言编写程序所需的时间的一小部分。MATLAB 名称代表矩阵实验室。MATLAB 最初的编写目的是提供对由 LINPACK 和 EISPACK 项目开发的矩阵软件的轻松访问,这两个项目共同代表了矩阵计算软件的最新技术。MATLAB 经过多年的发展,得到了许多用户的投入。在大学环境中,它是数学、工程和科学入门和高级课程的标准教学工具。在工业领域,MATLAB 是高效研究、开发和分析的首选工具。MATLAB 具有一系列称为工具箱的特定于应用程序的解决方案。对于大多数 MATLAB 用户来说非常重要,工具箱允许您学习和应用专业技术。工具箱是 MATLAB 函数(M 文件)的综合集合,可扩展 MATLAB 环境以解决特定类别的问题。可用工具箱的领域包括信号处理、控制系统、神经网络、模糊逻辑、小波、仿真等。